IgA Nephropathy: Hard Truths and Breakthrough Treatments — 4 New Options Patients Should Know
What is IgA nephropathy, and why does it drive CKD?
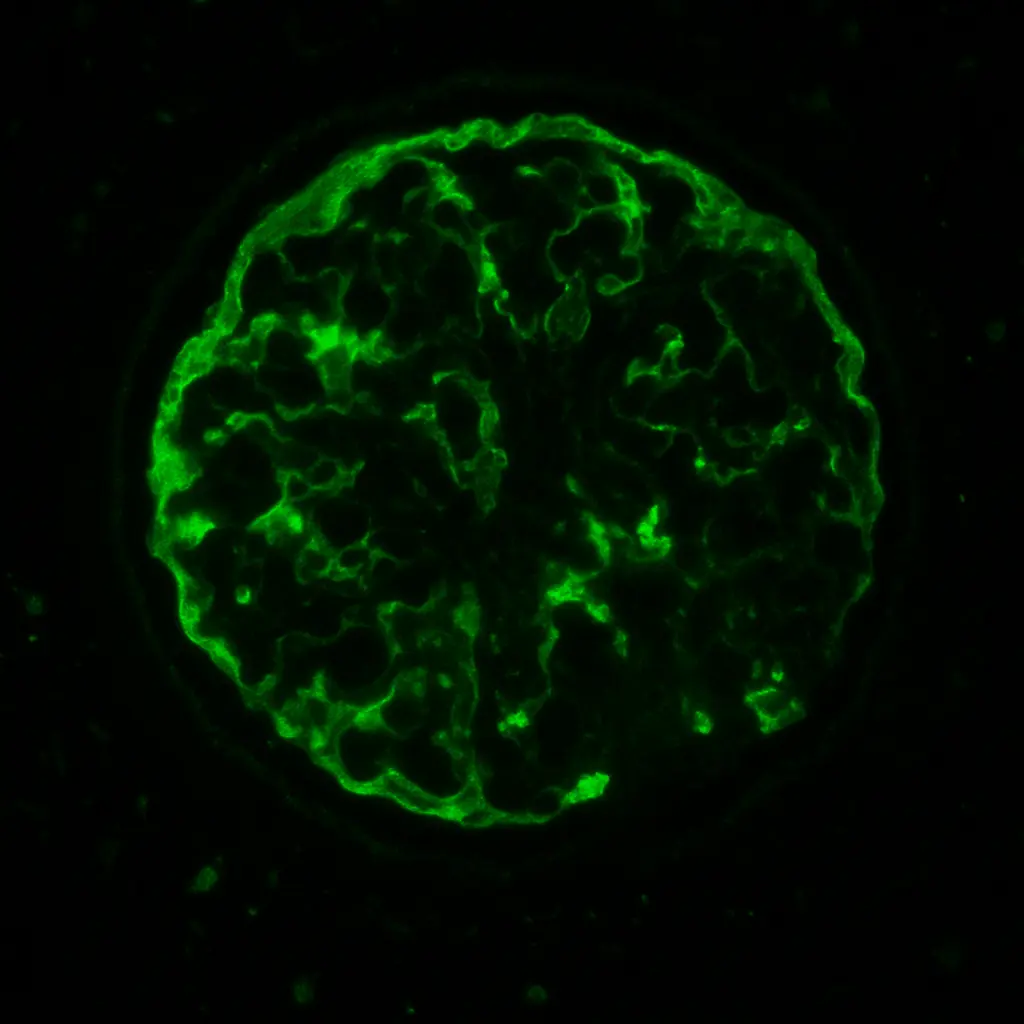
IgA nephropathy is one of the most common primary kidney diseases worldwide. At its core, it begins with an abnormal immune signal: the body produces galactose-deficient IgA1, which deposits in the glomeruli. These immune complexes set off inflammation and scarring, damaging the kidney’s delicate filtration system. Over time, protein leaks into the urine, blood pressure rises, and kidney function begins to slip away. For decades, patients with IgA nephropathy faced the same bleak story—supportive care only, with little hope of halting progression to kidney failure.
What triggers IgA nephropathy?
The spark behind IgA nephropathy lies in the immune system’s first line of defense: the mucosa of the respiratory and gastrointestinal tract. For reasons not fully understood-but likely a blend of genetics and environment-the body produces galactose-deficient IgA1 (Gd-IgA1), an antibody with a subtly altered sugar structure. Instead of protecting the body, these abnormal IgA molecules become targets. The immune system creates autoantibodies against them, which bind together in circulating immune complexes.
Once formed, these complexes have a particular destination: the glomerular mesangium. There, they lodge in the kidney’s filters, igniting complement activation (via the alternative and lectin pathways), mesangial cell proliferation, and inflammatory signaling. The clinical fallout is proteinuria, hematuria, and a slow but relentless decline in filtration capacity.

What sets IgA nephropathy apart is its tendency to flare with mucosal immune triggers-especially upper respiratory or gastrointestinal infections. This is why patients often notice “synpharyngitic hematuria,” where urine turns red or brown during a sore throat, cold, or GI illness. Each flare is not just a nuisance; it is a small assault on the kidneys that accumulates over time, ultimately shaping the course of chronic kidney disease.
The baseline that still matters: RAS blockade, BP control, diet, and SGLT2 support
Even with new therapies, the old cornerstones remain vital. Renin–angiotensin system (RAS) blockade with ACE inhibitors or ARBs is still first-line therapy, not because it “fixes” IgA nephropathy but because it reduces proteinuria and slows scarring. Lifestyle adjustments-blood pressure targets under 130/80, limiting salt, adopting structured diets like the CKD-DASH diet for kidney protection – are foundational. More recently, SGLT2 inhibitors have entered the supportive armamentarium, reducing proteinuria even in patients without diabetes. These therapies form the bedrock, upon which the new era of targeted options now stands.
New Option #1 – Targeted-release budesonide (Tarpeyo®): mucosal immune reset
Unlike systemic steroids that blanket the body with side effects, targeted-release budesonide acts in a more targeted manner. Its capsule design delivers the drug to the ileum, where Peyer’s patches generate much of the faulty IgA response. By acting locally, budesonide reduces abnormal IgA production at the source while minimizing systemic exposure.
The clinical signal was targeted: patients showed sustained reductions in proteinuria and a slower decline in eGFR, leading to FDA full approval in December 2023. Importantly, this is not indefinite therapy; typical courses last 9 months, and the benefit extends beyond discontinuation. But it is not benign-blood sugar can rise, infections can flare, and bone density requires monitoring. Still, for the first time, patients with IgA nephropathy had a targeted immune therapy that felt designed specifically for them.
New Option #2 – Sparsentan (FILSPARI®): dual-acting, non-immunosuppressive proteinuria control
The second breakthrough takes a completely different tack. Sparsentan combines dual endothelin A receptor antagonism with angiotensin receptor blockade in a single molecule. This double hit reduces intraglomerular pressure and calms fibrotic signaling, which translates into powerful proteinuria reduction. This drug requires discontinuation of existing ARB or ACE-inhibitor classes as these benefits are included with Sparsentan.
The PROTECT trial pitted sparsentan against irbesartan and demonstrated a significantly greater drop in proteinuria. By 2024, the FDA granted full approval, positioning sparsentan as a new first-line option for many patients. Unlike budesonide, sparsentan is not an immunosuppressant; instead, it modifies hemodynamics and cell signaling. The main caution lies in liver monitoring and pregnancy restrictions, which prompted a REMS program. This program requires eligible patients have scheduled liver function testing to monitor for complications. A 2025 update streamlined the REMS requirements, making sparsentan more accessible to community nephrology practices.
New Option #3 – Atrasentan (Vanrafia®): selective ETA antagonism with ALIGN data
The newest arrival, atrasentan, gained accelerated FDA approval in April 2025. Unlike sparsentan’s dual action, atrasentan is a selective endothelin A receptor blocker. The ALIGN trial showed clinically meaningful proteinuria reduction in patients with IgA nephropathy, enough to justify approval while outcome data continue to mature.
Where does atrasentan fit when sparsentan already exists? For some, it’s more selective action may translate into different tolerability, particularly regarding blood pressure and edema. It may also become a valuable add-on for patients not reaching proteinuria goals despite RAS blockade, SGLT2 therapy, and sparsentan. The real-world positioning of atrasentan will evolve quickly as payers, prescribers, and guidelines respond. This drug is an add on to existing use of ARB or ACE-i agents.
New Option #4 – Sibeprenlimab (Voyxact®): upstream APRIL blockade of IgA production
The newest therapeutic frontier targets the disease even earlier in its pathway: the overactive B-cell machinery that generates galactose-deficient IgA1. APRIL (A Proliferation-Inducing Ligand) is a key survival signal for plasma cells involved in this abnormal IgA production. By blocking APRIL, this agent reduces the upstream creation of pathogenic IgA before it ever reaches the kidney.
The FDA granted approval in late 2025 after trials showed substantial and durable reductions in proteinuria, accompanied by stabilization in eGFR across a broad range of risk profiles. Unlike budesonide, which modulates mucosal immunity, or sparsentan and atrasentan, which act on hemodynamics, APRIL inhibition reshapes the immune engine at its source. Patients may see reduced immunoglobulin levels over time, making infection monitoring essential, and vaccine timing may require coordination.
Where does APRIL blockade fit in the sequence of care? For some, it may become the preferred option when mucosal-directed therapy is insufficient or when endothelin antagonists are not tolerated. For others, it may serve as an escalation step after sparsentan or atop standard-of-care regimens. As with every new IgA therapy, real-world positioning will refine rapidly, but the arrival of APRIL inhibition marks a true shift: an immune-targeted therapy with upstream precision, designed for the biologic heart of IgA nephropathy itself.
Where SGLT2 inhibitors fit in IgA nephropathy
SGLT2 inhibitors deserve their own mention. Subgroup analyses from DAPA-CKD revealed that dapagliflozin reduced progression risk in patients with IgA nephropathy, even without diabetes. KDIGO’s 2024 draft guidelines acknowledged this, placing SGLT2 inhibitors alongside RAS blockade as foundational therapy. Importantly, these drugs layer well with the new targeted agents. A future care pathway may routinely include RAS blockade, SGLT2 inhibitor, plus either sparsentan, atrasentan, or budesonide-sequenced according to proteinuria levels, eGFR thresholds, and patient tolerance.
For readers seeking more context: see Proteinuria: why it matters
KDOQI’s U.S.-Focused Take on KDIGO’s IgA Nephropathy Guidance
In early 2025, the KDOQI work group released its commentary on the KDIGO 2024 IgAN guideline, tailoring the recommendations for U.S. practice. They endorsed KDIGO’s shift toward early disease-modifying therapy in parallel with standard supportive care, rather than waiting for proteinuria or eGFR decline to worsen. They also stressed the central role of SGLT2 inhibitors as part of foundational care across CKD etiologies, including IgA nephropathy.
Importantly, KDOQI raised the point that this evolving therapeutic landscape will likely reshape thresholds for kidney biopsy in patients presenting with proteinuria. Historically, a biopsy was pursued mainly when proteinuria exceeded 3 g/day or renal function showed unexplained decline. With targeted therapies now tied directly to histologic confirmation, nephrologists may need to consider biopsy earlier in the course of unexplained proteinuria, even at sub-nephrotic levels, to secure a definitive diagnosis and justify access to high-cost novel agents. KDOQI highlights that this change is not just clinical but also systemic-insurance approval and payer policy will increasingly depend on biopsy-proven disease
Who qualifies for these therapies? Quick criteria and red flags
- Therapeutic target: The contemporary goal is to drive proteinuria down to <500 mg/day (PCR <0.5 g/g), since outcomes improve most dramatically below this point.
- eGFR floors: Budesonide has been studied down to eGFR ~35; ERAs can extend lower but require caution.
- Pregnancy: Both sparsentan and atrasentan are contraindicated.
- Monitoring: Budesonide → glucose, bone, infection risk; Sparsentan → liver function and fluid status; Atrasentan → fluid retention, hemoglobin.
Straight talk on risks, costs, and realistic expectations
These drugs are not cures. Proteinuria reduction is a surrogate marker for kidney protection; it forecasts slower CKD progression but doesn’t guarantee freedom from dialysis. Costs remain daunting-tens of thousands annually-and insurance approvals can be exhausting. Adverse effects, though more manageable than systemic steroids, still exist. For patients, the “hard truth” is that these new tools extend the runway, buying time and hope, but vigilance remains necessary.
How to talk with your nephrologist about next steps
Bring recent labs, especially proteinuria quantification and eGFR trends. Ask: Am I optimized on RAS and SGLT2 therapy? Am I eligible for budesonide, sparsentan, or atrasentan? What would we monitor if I start? Include your renal dietitian in the conversation-medications and nutrition are intertwined. Patients who engage actively in this dialogue, journaling their labs and blood pressure in tools like a CKD tracker, empower themselves in ways that medications alone cannot.
FAQs
Can these be combined? Early signals suggest layering SGLT2 inhibitors with ERA or budesonide is safe, but ERA combinations with each other are not advised.
How long until effect? Proteinuria reduction is often seen within months, but long-term kidney protection plays out over years.
What if my eGFR is <30? Therapies are less studied here, but individual judgment applies; discuss risks with your nephrologist.
Last updated: December 2025
Works Cited
- KDIGO 2024 Clinical Practice Guideline Draft for IgA Nephropathy and Vasculitis.
- Cattran DC, Appel GB, Bomback AS, D’Agati VD, Lafayette RA, Rovin BH, et al. KDOQI US Commentary on the KDIGO 2024 Clinical Practice Guideline for the Management of IgA Nephropathy and IgA Vasculitis. Am J Kidney Dis. 2025 Mar;85(3):291-304. doi:10.1053/j.ajkd.2024.11.003. PMID: 39556063.
- FDA approval announcements and package inserts for budesonide (Tarpeyo/Nefecon, 2023), sparsentan (FILSPARI, 2024), and atrasentan (Vanrafia, 2025).
- DAPA-CKD trial subgroup analyses.
- PROTECT and ALIGN trial publications.
- ENVISION and VISIONARY trial publications.